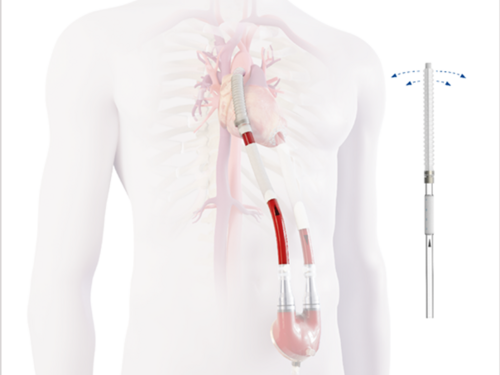
EXCOR® Graft Cannula: The new flexibility of graft anastomosis
This innovative new EXCOR® Graft Cannula was developed to offer the surgeons a wide range of applications and flexibility during surgery by combining the proven flow properties of EXCOR® cannulae with the advantages of a graft anastomosis.
Improvements in the field of mechanical circulatory support and the further development of ventricular assist devices enable critically ill heart patients to recover and bridge the often very long waiting periods for a donor heart. Berlin Heart has developed a cannula that intends to a more convenient surgical handling during implantation and an adaptation to the individual needs of patients of all ages treated with the EXCOR® VAD system, from newborns to adults.
The pediatric heart medicine team from the Universitätsklinikum Hamburg Eppendorf (UKE) in Germany was the first to implant this new outflow cannula. Prof. Dr. med. Michael Hübler about his first experience: “The new cannula was easy to assemble and implant. The flexible design helps the surgeon adapt to the individual physiology of patients.”
The soft graft material also has another very important advantage: It allows to reduce the anastomosis-angle and this leads to low flow disturbances with the aim to optimize hemodynamics.1
Dr. med. Ares K. Menon, Cardiac Surgeon and General Manager of Berlin Heart GmbH summarizes the project goal of this further development as follows: “With the EXCOR® Graft Cannula we satisfy the unmet clinical need for much more flexible and convenient cannulae for aortic and pulmonary cannulation of patients of all ages.”
Heart's philosophy is based on future orientation and investment in research and development, thereby always in focus to offer a suitable solution for every single patient.
Another important milestone in the history of Berlin Heart was reached with this new development.
1 Effect of LVAD outflow conduit insertion angle on flow through the native aorta, KD May-Newman, BK Hillen, CS Sironda & W Dembitsky Journal of Medical Engineering & Technology Vol. 28 , Iss. 3,2004
About Berlin Heart
Berlin Heart GmbH develops, manufactures, and markets innovative ventricular assist devices (VADs) for mechanical circulatory support. With the EXCOR® Adult and the EXCOR® Pediatric, Berlin Heart is the only company in the world able to provide support to patients of every age and size, from infants to adults, thereby meeting an important worldwide unmet need.
Berlin Heart’s systems provide left ventricular, right ventricular, or biventricular support (both sides); hospitals around the world use the system for short-, medium-, and long-term circulatory support, providing patients with a life-saving treatment option.
Users can also access round-the-clock medical and technical support.
Press Contact:
Berlin Heart GmbH
Marketing & Public Relations
Theresa Tolle
Wiesenweg 10
12247 Berlin
Tel. 030 8187 2600
theresa.tolle@berlinheart.de
Disclaimer: The access to some or all shown products may be restricted by country-specific regulatory approvals. The use of EXCOR® VAD for adults, RVAD-support, EXCOR® Venous Cannula, EXCOR® Arterial Cannula for Graft, Excor mobil and EXCOR® Active is not FDA approved and not available for commercial use in the US.